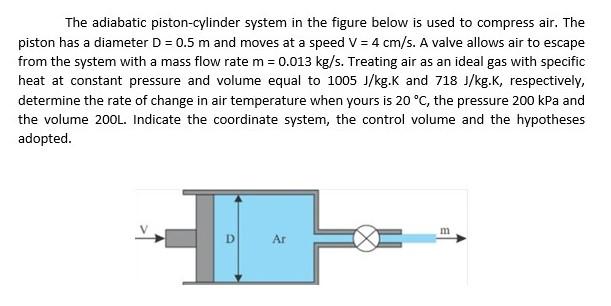
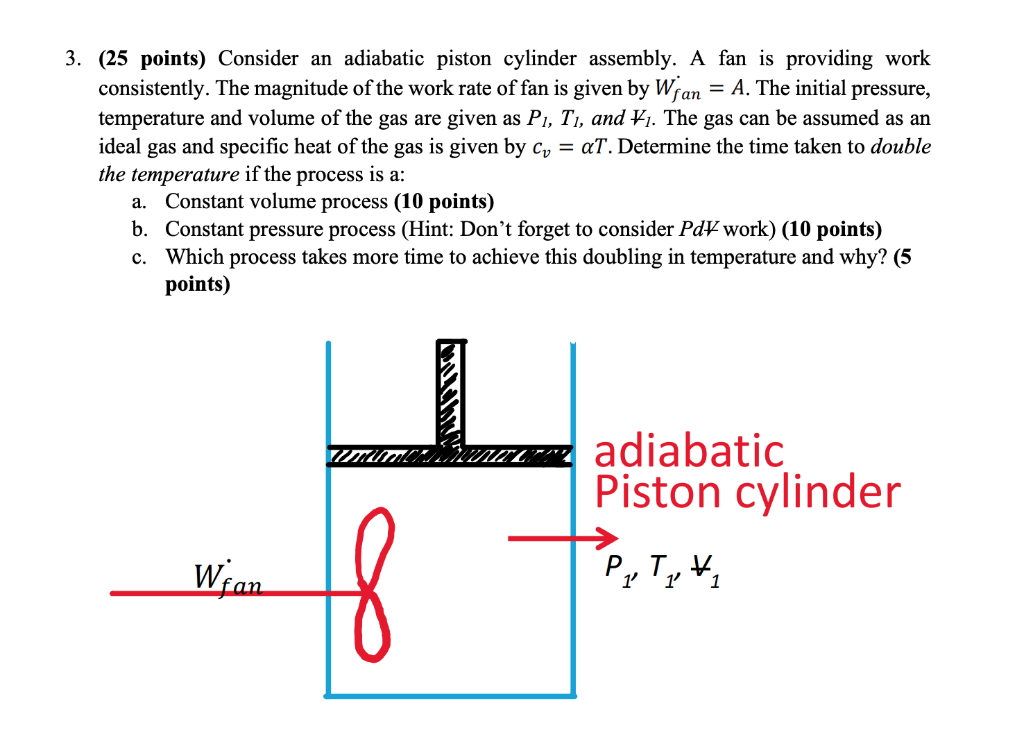
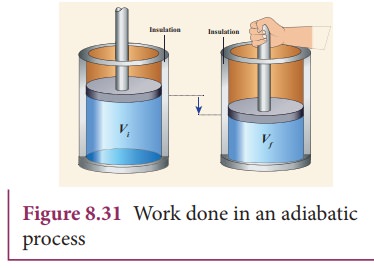
Ideal Adiabatic Process of A Piston Expanding (Find Work and Total Internal Energy Change) - YouTube

SOLVED: Consider a well-insulated (i.e., adiabatic) piston-cylinder assembly that goes through the following compression process (process C). The gas inside the piston-cylinder is filled with 1 mol of Ar gas, initially at

An adiabatic piston of mass m equally divides an insulator container of length l. One end of alight spring is connected to the piston and other end to the right wall. The
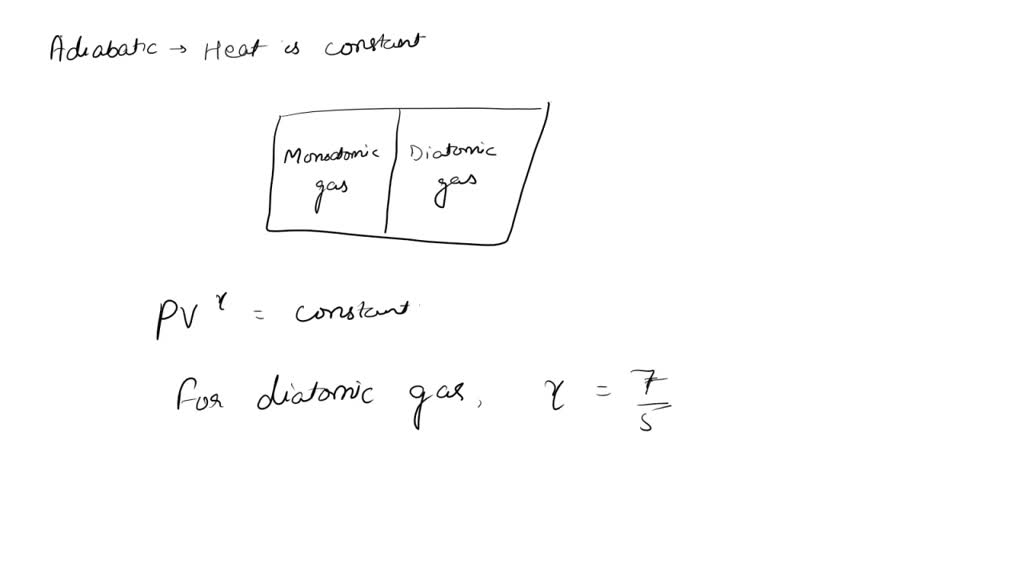
SOLVED: An adiabatic cylinder fitted with an adiabatic piston at the right end of cylinder, is divided into two equal halves with a monoatomic gas on left side and diatomic gas on




![PDF] From the adiabatic piston to macroscopic motion induced by fluctuations | Semantic Scholar PDF] From the adiabatic piston to macroscopic motion induced by fluctuations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a3ee3158617a875f430b330071b30ed116759605/2-Figure1-1.png)